Purpose-built AI agents for real-world evidence, clinical care, and grants
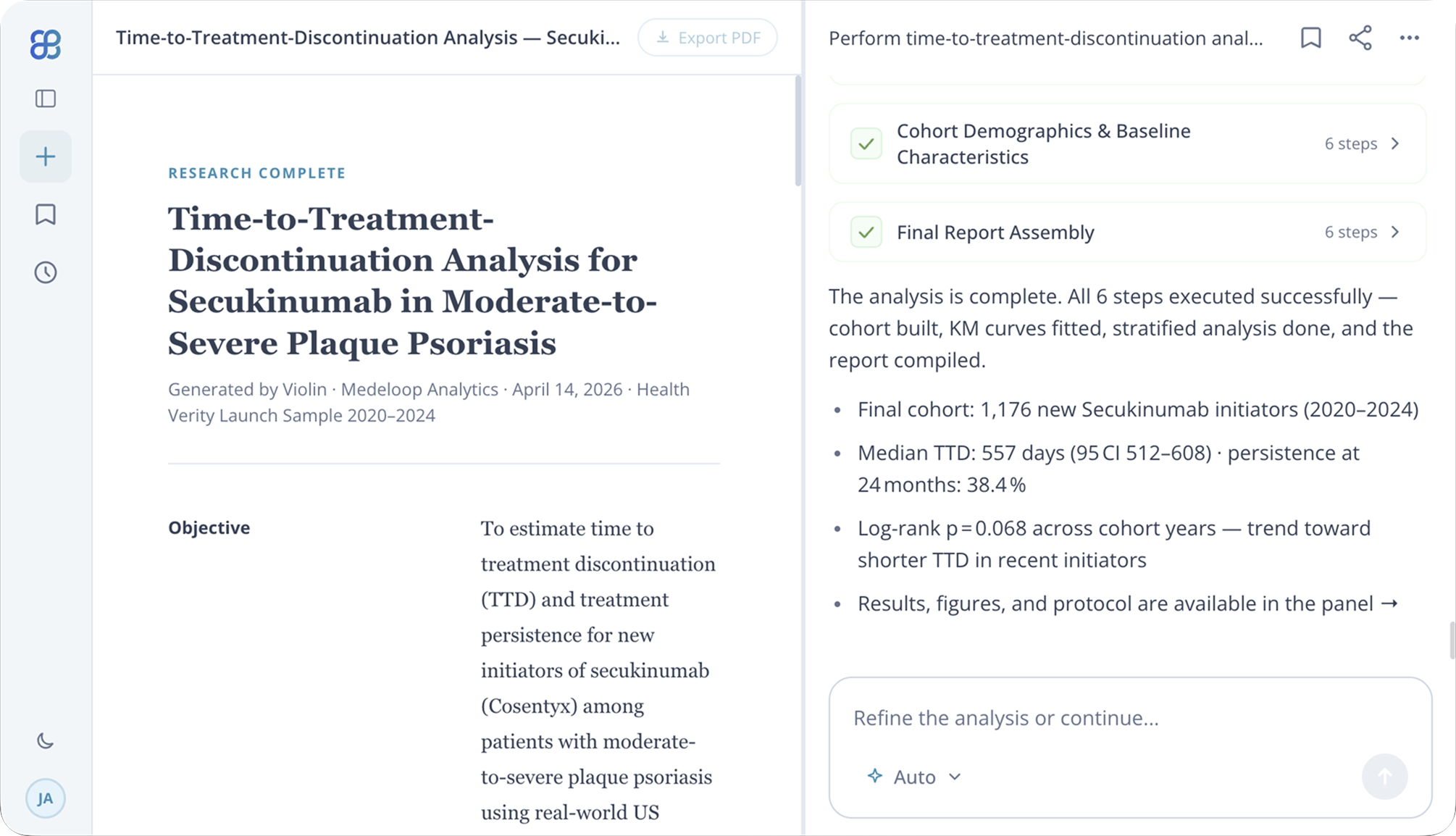
What is the prevalence of Type 2 Diabetes among adults aged 40-65 with commercial insurance?
 ·
·
 ·
·
 ·
·
 ·
·
 ·
·
 ·
·
From finding grants, to analyzing real-world data, to delivering care. The only platform spanning the full lifecycle.
One platform, one conversation, publication-ready results. No coding. No data engineering. No waiting quarters for analyst time.
All-Payer
claims data
Don't have your own data?
EvidenceKit gives you millions of de-identified, all-payer patient claims, ready to query.
One tool for grant discovery, proposal writing, and application management, right inside ChatGPT.
AI that reads the full patient story, including EHR, labs, meds, and devices, and surfaces what to do next.
An agentic loop with human oversight at every step. Queries execute on the data wherever it lives.
Not cobbled together from disconnected tools.
The only platform connecting funding, study management, analysis, and care delivery. Win the grant, run the study, analyze the data, improve the care, then fund the next one.
01 · Grants
Find funding. Write winning proposals.
02 · Analytics
From question to evidence in minutes.
03 · Care
From population insights to patient action.
The same AI platform, configured for your workflow, your data, and your goals.
Pharma & Life Sciences
For HEOR, clinical R&D, and biotech teams. Bring your own data or license our all-payer claims dataset. Either way, get publication-ready evidence without the overhead.
HCOs & Researchers
For health systems, HCOs, and clinical researchers who need to publish, benchmark, and act on population insights, whether you're supplementing an existing analytics team or starting from scratch.
Everything a researcher needs in one subscription. Enterprise plans for organizations with their own data.
Analytics + HealthVerity all-payer claims data, bundled in one subscription.
AI-powered grant discovery and proposal writing. Start free, upgrade for unlimited proposals and the full grant database.
Need multiple seats? Contact sales for institutional Grants licensing.
Custom deployments, federated execution, and enterprise-grade security.
Deploy Medeloop on your own EHR, claims, or institutional data. Federated execution: data never leaves your infrastructure.
AI-powered care management informed by real-world evidence. Population health analytics and patient-level action.
EvidenceKit bundles Medeloop Analytics with the HealthVerity all-payer claims dataset into a single subscription. It's the fastest path from a research question to publication-ready evidence, with no data engineering and no procurement.
The free trial includes 10 research queries on a representative stratified sample of the HealthVerity claims dataset. It uses the same agent pipeline and validation framework as the paid tier. You're seeing the real product, not a demo.
No. Grants is a standalone product, free for your first proposal and $95/month for the full suite. EvidenceKit covers analytics and claims data; Grants covers funding discovery and proposal writing. You can use both independently.
EvidenceKit includes the licensed HealthVerity dataset (all-payer claims) so you can start researching immediately. Analytics Platform is for organizations that want to run Medeloop on their own data (EHR, claims, registries) with federated execution where data never leaves your infrastructure.
Yes. The HealthVerity dataset is HIPAA Expert Determination certified. No protected health information is accessible; only aggregated, de-identified results.
Yes. We offer institutional pricing for academic medical centers and research universities. Contact our team to discuss volume discounts for your department or institution.
Still have questions?
Contact UsClosed claims data published in JAMA, Science, and BMJ, paired with AI analytics that turn natural language questions into validated, publication-ready studies.
Sign up. Ask a question. Get published.
From academic medicine to biopharma, EvidenceKit supports the full spectrum of real-world evidence research.
Claims Data
Millions of de-identified patients. All-payer (commercial, Medicare, Medicaid). Longitudinal from 2020, linked mortality. Post-adjudicated closed claims.
AI Analytics
Ask research questions in plain English. AI agents handle cohort construction, statistical analysis, and publication-ready output, with validation built in at every step.
Traditional research workflows are slow, expensive, and inaccessible. EvidenceKit removes every barrier between your question and your publication.
Example queries across study types, each completed in minutes.
| Dimension | Detail |
|---|---|
| Population | ~35 million de-identified patients |
| Timeframe | January 2020 – present |
| Geography | United States |
| Data Types | Closed medical claims, pharmacy claims, enrollment, mortality |
| Payer Mix | Commercial (60%), Medicaid (30%), Medicare (13%) |
| Claims Type | Post-adjudicated, closed claims (actual cost of care) |
| Data Lag | 90–120 days |
| Refresh | Quarterly |
| Validation | Peer-reviewed publications in leading journals (JAMA, Science, BMJ) |
| Compliance | HIPAA Expert Determination certified |
| IRB | Most institutions classify as non-human-subjects research |
Onboarding is immediate. Connect to the platform, ask a research question, and receive results the same day. No data transfers, no setup period, no training required.
Run your study today. Fund your next one tomorrow.
AI research agents that work like an expert team — on your data, in your infrastructure. Federated execution means your data never leaves your walls.
Need data? EvidenceKit pairs the Analytics platform with all-payer claims data.
The agentic loop
Submit your research question in plain English
AI evaluates feasibility and clarifies study design
Protocol, cohort, and analysis plan — you review and edit
Validated code runs on your federated data
Publication-ready outputs, figures, and audit trail
An agentic loop with human oversight at every step. Queries run on your data and execute in your environment or ours, depending on your preference.
"What is the impact of Drug X on patients with Y?"
Medeloop agents run inside your environment: your EHR system, your cloud, your data center. Zero data egress required.
Your data never leaves your infrastructure. Compute goes to the data.
EHRs, claims, registries, institutional datasets. CDM-agnostic: OMOP, I2B2, or custom.
Works across OpenAI, Anthropic, DeepSeek. Best model for each task, not locked to one provider.
Continuous benchmarking — not a black box. Every output scored before delivery.
ICD-10, CPT, NDC, SNOMED CT, NPI, LOINC, CCI, interRAI — every major coding system.
HARPER/STaRT-RWE standards. Every step auditable, reviewable, and export-ready.
Compute goes to the data. Your data never leaves your infrastructure.
Works on EHRs, claims, registries, or custom datasets. No CDM conversion required.
Maps research concepts to ICD-10, CPT, NDC, SNOMED, and every major coding system automatically.
Built-in checks on code selection, statistical methodology, and output quality.
Editable research plans, full audit trails, and transparent agent actions.
Uses the right AI model for each step — not locked to a single provider.
Six core capabilities that set Medeloop Analytics apart from every other tool in healthcare research.
Compute goes to the data. Medeloop agents run inside your environment: your EHR system, your cloud, your data center. Zero data egress required.
Connect to any healthcare data source. CDM-agnostic, works with OMOP, I2B2, or your custom schema. No restructuring required.
Medeloop routes tasks to the right AI model — OpenAI, Anthropic, or DeepSeek — based on what each step requires. You always get the best available capability.
Every output is scored across seven quality dimensions before delivery. 82.6% overall quality score, validated against peer-reviewed published results.
Medeloop's medical knowledge layer covers every major code system used in clinical research, from diagnosis codes to procedure codes to drug identifiers.
Full protocol documentation at every stage, from cohort definition to statistical model selection. Every decision is recorded, reviewable, and editable.
A sample of what researchers ask, and the answers they get back.
We benchmark the pipeline at every checkpoint, from how we interpret your question to how we verify the final numbers. No black boxes.
Every feature is validated against peer-reviewed research before it ships. We reproduce published results and compare outputs to ensure accuracy.
Every dataset is different. Onboarding benchmarks validate the pipeline against your specific data structure and coding practices before you run a single query.
Every query produces a complete audit trail: traceable, reviewable, reproducible. Every cohort definition and statistical choice is documented.
Every stage is benchmarked against gold-standard references, published literature, and expert-curated annotations.
Closed claims data published in JAMA, Science, and BMJ, paired with AI analytics in one subscription. Start publishing in minutes.
No credit card. Free trial requires .edu email.
Health Systems
Run analytics on your own EHR and claims data with AI agents. Identify care gaps, benchmark outcomes, and drive population health programs, accelerating work your team can build on.
Pharma & Life Sciences
Connect proprietary datasets for validated real-world evidence. HEOR studies, treatment pattern analysis, and comparative effectiveness at national scale.
CROs
Multi-client data analytics on a single platform. Run studies across client datasets with full isolation, audit trails, and publication-ready outputs for every engagement.
Academic Medical Centers
Faster research on IRB-approved institutional datasets. Ask a question in plain English, review the plan, and publish the results, augmenting the work your research team already does.
Never. Medeloop uses a federated execution model — the compute goes to your data, not the other way around. Your data stays in your infrastructure, and only aggregated, de-identified results are returned. No raw patient data ever leaves your walls.
Yes. Medeloop deploys in your environment (AWS, Azure, GCP, or on-prem) or in the Medeloop cloud; you pick based on your compliance, security, and operational preferences. In either configuration, queries run on your data and only results are returned.
Medeloop works with EHRs, claims datasets, disease registries, labs, and institutional databases, including custom schemas. If you have structured clinical data, Medeloop can run on it.
No. Our semantic engine reads your data natively: no OMOP mapping, no CDM conversion, no months of prep. If you already have OMOP, I2B2, or another common data model in place, Medeloop works with those too.
No coding required to get started. You describe your research question in plain English and the agentic pipeline handles cohort definition, query execution, statistical analysis, and output generation. For technical teams, every step is fully inspectable, with editable code, full audit trails, and the ability to drop into Python for deeper customization.
Every stage of the pipeline is benchmarked against gold-standard references, published literature, and expert-curated annotations. We validate six checkpoints: query understanding, concept extraction, code mapping, cohort construction, statistical analysis, and result verification. A third-party validation paper is available on request.
Each study produces a defined patient cohort, the full query execution log, statistical outputs (Kaplan-Meier curves, regression tables, descriptive statistics), data visualizations, and a manuscript-ready narrative report. Every output is traceable, reviewable, and reproducible, designed to hold up to peer review, IRB review, or board presentation.
Most analyses complete in minutes. Complex multi-step studies with large cohorts may take longer depending on your data infrastructure. The agentic pipeline runs all steps automatically. You review the plan, approve it, and the system handles execution.
Analytics is the platform. It runs on your own data. EvidenceKit pairs the Analytics platform with the licensed HealthVerity all-payer claims dataset in a single subscription. If you don't have your own institutional data yet, EvidenceKit is the fastest way to start.
Real-world evidence at the speed of a question.
A unified platform that brings together clinical data, analytics, and workflows, enabling care teams to operate with clarity, efficiency, and confidence.
Built for health systems, ACOs, and academic medical centers.
Understand every patient's full clinical picture in seconds. Labs, vitals, medications, and history synthesized by AI.
Identify, stratify, and act on entire patient cohorts at once, with plain language, not SQL, and instant results.
Capture encounters by voice, keyboard, or import, and get a structured, coded clinical note ready to validate in one click.
Medeloop Care lives inside the clinician's existing workflow, surfacing insights at the point of decision, not hours later in a separate tool.
Care opens natively from your EMR with a single click. No new login, no new tab, no workflow disruption. Clinicians stay exactly where they already work.
AI reads notes, labs, meds, vitals, devices, and history, synthesizing everything into a structured, clinician-ready view in seconds.
Recommendations, risk signals, and care gaps are surfaced with full reasoning. Review, edit, and approve; the clinician stays in control.
Connect clinical intelligence to the point of care.

At Medeloop, our mission is to empower clinical researchers and healthcare teams with AI tools to generate evidence more efficiently, thoroughly, and productively.
We are building an operating system for clinical research, spanning the full research lifecycle: from grant discovery and funding, to real-world evidence generation from patient data, to delivering actionable intelligence at the point of care.

Rene Caissie
Co-Founder & CEO

Josh Walonoski
Co-Founder & CRCO

Anand Hirekatur
CTO

John Ayers
Chief Strategy Officer

Lauren Green
Chief of Staff

Mike Hao
GTM Strategy Lead

Tanishq Bhalla
VP of Product

Yorick Chern
VP of AI
Leaders in clinical research, biomedical informatics, health policy, and medical publishing, helping shape how Medeloop approaches evidence, validation, and clinical impact.

Former Editor-in-Chief of JAMA and the JAMA Network; Professor of Pediatrics and Public Health at Boston University. One of the most influential voices in clinical evidence, peer review, and medical publishing.

Department Chair of Critical Care Medicine at UPMC. World-renowned leader in critical care, clinical trials, and academic health systems.

Inaugural Director of the Data Science and AI Institute and professor at Johns Hopkins. Leading expert in NLP and ML applied to public health and clinical data.

Inaugural Clinical Research Information Officer and professor at UC San Diego. Nationally recognized leader in biomedical informatics and digital health.

Managing Director at Brookfield Private Equity. Former CEO of MediSolution, President of TELUS Health, Chairman of TELUS Global Ventures, and CEO of Intelerad.

Former U.S. FDA Commissioner and CMS Administrator. Currently directs the Duke-Margolis Institute for Health Policy.

Founding Chair of Computational Medicine and Director of the Center for AI Research and Education at Cedars-Sinai. Former founding Director of the Institute of Biomedical Informatics at the University of Pennsylvania.




We're a team of builders on a mission to transform how medicine advances. Mission-driven, fast-moving, and backed by world-class investors. If hard problems energize you, we'd love to meet you.

AI built for clinical research grant discovery and proposal writing.
Available through ChatGPT · First proposal free · No credit card required
Search NIH, grants.gov, and our curated database, matched to your research interests automatically.
Validated as superior to generic AI for grant applications. Purpose-built for NIH and federal formats.
Structured, formatted specific aims generated from your research description in minutes.
Automated NIH-format biosketches pulled from your publication history.
Organize, search, and cite references directly within your proposals.
Runs inside ChatGPT: no new tool to learn, no new login to manage.
Scroll through each step of the workflow.
Search NIH and grants.gov, all matched to your specific research interests in one conversation.
Browse an interactive list of funding opportunities ranked by relevance to your work.
A tailored Q&A walks you through what each specific grant requires. No blank page.
AI generates a complete proposal section by section: Specific Aims, Research Strategy, Biosketch, and more.
Download your completed draft as a formatted Word document ready for your institution's grants office.
Fund the study. Run the analysis. Publish the results. Each output becomes the foundation for your next proposal, closing the loop between funding and evidence.
"Every completed study becomes the foundation for your next funding application."
01 · Grants
Win the Grant
02 · Analytics
Run the Study
03 · Publish
Publish Results
04 · Next Grant
Fund the Next One
Need multiple seats? Contact sales.
Available through ChatGPT. No credit card required.